OtoSet by SafKan Health Introduces First Automated and FDA Cleared
Par un écrivain mystérieux
Description
SEATTLE, WASHINGTON — Having recently announced their partnership, SafKan Health is excited to provide Beltone‘s nationwide network with the opportunity to offer patients an innovative ear cleaning service to manage excessive earwax. According to the American Academy of Otolaryngology, excessive earwax is present in over 35 million Americans including 1 in 10 children, 1 in 20 adults, and more than one-third of the senior population.

OtoSet® (@safkanhealth) • Instagram photos and videos

SafKan Health announces FDA 510(k) clearance for OtoSet - Medical Buyer

Brothers behind SafKan Health tackle wax with ear-cleaning headset - Puget Sound Business Journal

Nicole Denney, Au.D. on LinkedIn: #otoset #thaa #earwaxremoval #cerumenmanagment #ears

SafKan Health

SafKan Health OtoSet® FDA-Cleared Earwax Removal Device

FDA-Cleared OtoSet® Ear Cleaning System (Dr. Tracy Board - Audicles Hearing Services)

SafKan Health OtoSet® FDA-Cleared Earwax Removal Device

Johari Digital manufactures SafKan Health's revolutionary product OtoSet®- The First Automated & FDA Cleared Ear Cleaning Device
OtoSet® by SafKan Health Introduces the First Automated & FDA Cleared Ear Cleaning Device to Manage Earwax Buildup
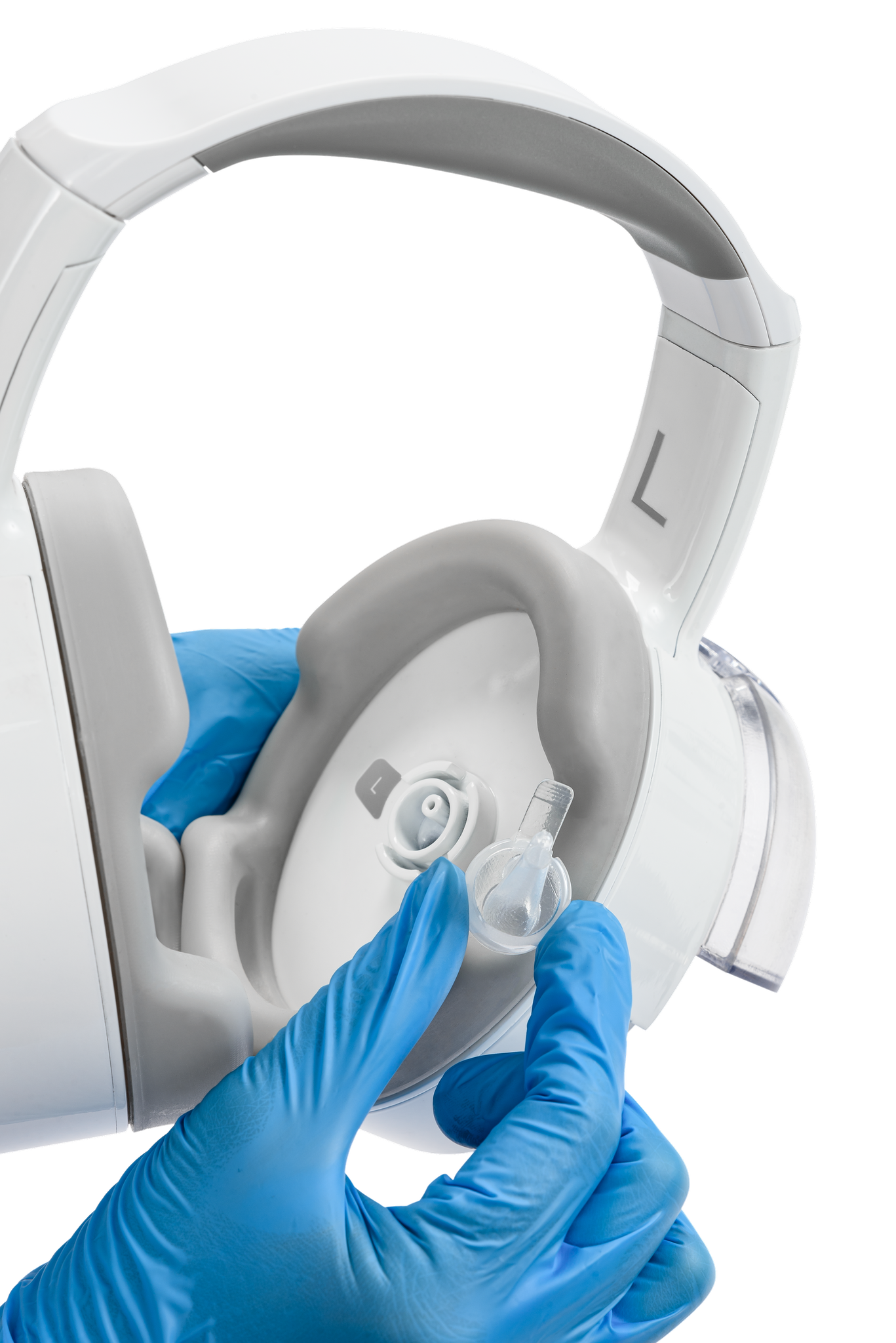
SafKan Health OtoSet® FDA-Cleared Earwax Removal Device
depuis
par adulte (le prix varie selon la taille du groupe)




-030247-pa.jpg)


